Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (10): 1573-1579.doi: 10.3969/j.issn.2095-4344.0718
Previous Articles Next Articles
Effects of titanium implant surface modification on macrophage polarization and osteogenesis
- Hospital of Stomatology, Tianjin Medical University, Tianjin 300070, China
-
Received:2018-01-23Online:2018-04-08Published:2018-04-08 -
Contact:Li Chang-yi, Professor, Doctoral supervisor, Chief physician, Hospital of Stomatology, Tianjin Medical University, Tianjin 300070, China -
About author:Li Ying, M.D., Associate chief physician, Master’s supervisor, Hospital of Stomatology, Tianjin Medical University, Tianjin 300070, China -
Supported by:the National Natural Science Foundation of China, No. 31470920, 81500886; the Natural Science Foundation of Tianjin, No. 16JCYBJC28700; the Natural Science Foundation of Hebei Province, No. E2017202032
CLC Number:
Cite this article
Li Ying, Li Chang-yi.
share this article
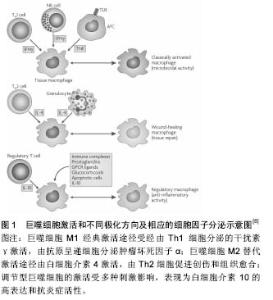
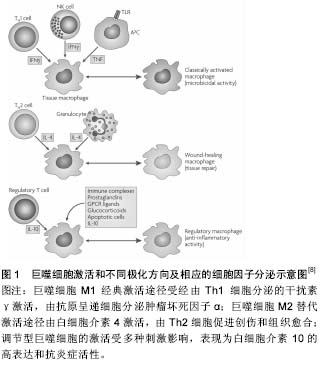
2.1 巨噬细胞的极化和诱导成骨特性 2.1.1 巨噬细胞的极化 巨噬细胞是天然免疫的重要组成部分,具有异质性和可塑性,在维持内环境稳态、免疫调控过程中发挥着关键作用[5]。如图1所示:巨噬细胞通常被分为促炎症的经典活化途径巨噬细胞(M1型)和抗炎症的替代活化途径巨噬细胞(M2型)。经典的M1型极化由幼稚巨噬细胞经由辅助性T细胞1(Th1)分泌的干扰素γ和革兰阴性菌脂多糖激活,属于促进炎症反应,通常发生于微生物或其他急性威胁状态下的快速免疫激活[6-7],以高水平的白细胞介素1β、白细胞介素6、肿瘤坏死因子α和诱导型一氧化氮合成酶表达为特 征[6-11]。M2激活属于创伤愈合和组织重建反应,其标志为抗炎细胞因子白细胞介素10、白细胞介素4、白细胞介素13和转化生长因子β的表达[8,12]。M2替代途径激活时的表面标志物为精氨酸酶1、CD163和CD206,辅助增强Th2,下调炎症,促进骨愈合[12]。除了M1和M2基因型之外,还有另一种巨噬细胞基因型,其特征是白细胞介素10的高表达,而且可被称为去激活或调节型的巨噬细胞[13]。单核细胞/巨噬细胞作为人体中所发现的最丰富的细胞类型之一,在骨-生物材料整合中起着关键性作用。"
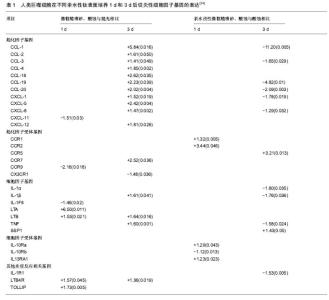
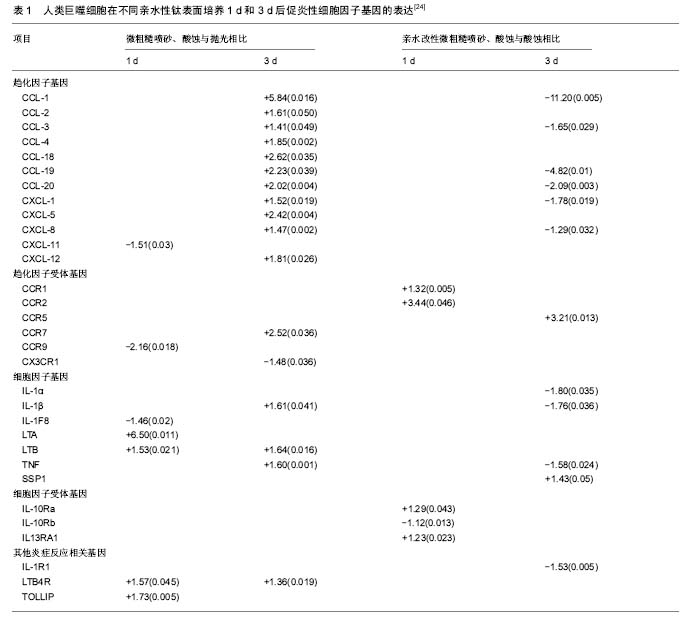
2.1.2 巨噬细胞的诱导成骨作用 种植体成功的骨结合由炎症过程驱动[14],巨噬细胞在炎症期分泌多种细胞因子和递质,包括促炎症细胞因子(肿瘤坏死因子α)和抗炎症递质(白细胞介素10),以及某些生长因子(转化生长因子β)。适度的炎性细胞因子可促进骨折愈合,而过度的炎症不利于骨重建和骨折愈合,通过调节巨噬细胞极化,控制炎症反应对于正常的骨修复和内稳态平衡是至关重要 的[15]。此外,骨组织中常驻的骨巨噬细胞在调节骨形成、骨折和骨伤口愈合方面也发挥重要作用[3-4,16]。巨噬细胞产生的抑瘤素M被鉴定为特异性巨噬细胞因子,支持人类成骨细胞的成骨细胞潜能[17]。总之,巨噬细胞在各种形式的骨形成中均发挥重要的诱导成骨作用。 2.2 种植体表面改性对巨噬细胞极化的影响 2.2.1 种植体表面物理、化学性质对巨噬细胞极化的影响 种植体表面纳米形貌对巨噬细胞的影响:胶体刻蚀法精确制备的纳米形貌种植体周围,可见降低炎症细胞因子肿瘤坏死因子α、破骨细胞标志物CatK的基因表达和CD163阳性巨噬细胞,骨膜素免疫染色增高,从而增强矿化、减弱炎症反应[18]。表面受控的纳米形貌叠加螺旋形钛种植体植入大鼠胫骨后,纳米图案下调单核细胞趋化蛋白1表达,上调骨钙素的表达,更多新的类骨质和编织骨形成,纳米形貌减弱了炎症过程,增强了骨整合早期成骨反应[19]。纳米形貌对于巨噬细胞的影响与纳米结构尺寸孔径相关。具有相似尺寸范围(70-80 nm)的纳米管减少肿瘤坏死因子α、白细胞介素1β、单核细胞趋化蛋白1、白细胞介素6和巨噬细胞炎性蛋白1α在原代人单核细胞/巨噬细胞和巨噬细胞系中的表达[19-20];而Ma等[21]发现,不同管径的纳米管形貌诱导不同方向的巨噬细胞极化,其中 30 nm直径纳米管诱导愈合相关的M2极化,而80 nm直径的纳米管种植体表面则诱导促炎M1方向的巨噬细胞极化。具有相似纳米尺寸范围但具有凹槽形状的表面刺激巨噬细胞RAW264.7中肿瘤坏死因子α和白细胞介素1β的表达/分泌。无论是大小(微米对纳米)还是形状(例如管对凹槽)都是巨噬细胞激活的重要决定因素。 纳米改性种植体表面限制巨噬细胞通过黏附、活化和融合的方式向异物巨细胞的转变。种植体表面的平行纹路,直接影响巨噬细胞的形态和相关细胞因子分泌,导致无法正常黏附,诱导凋亡,减轻免疫反应程度,促进伤口愈合和骨整合[22]。非极化(M0)或极化巨噬细胞(M1和M2)产生的不同条件培养基对骨髓间充质干细胞有独特的作用,种植体植入后对巨噬细胞极化的精细调控可能是调节骨髓间充质干细胞治疗潜力的有效方 法[23]。综上所述,纳米形貌对于巨噬细胞的影响因素复杂,尚有待进一步深入研究。 种植体表面亲水性和粗糙度对巨噬细胞的影响:Alfarsi等[24]将人类巨噬细胞系暴露于3种不同亲水性的钛表面(抛光后,微粗糙喷砂、酸蚀,亲水改性微粗糙喷砂酸蚀)后,检测84种细胞因子、趋化因子及其受体的基因表达情况。由表1可见,微粗糙喷砂、酸蚀表面引起肿瘤坏死因子、白细胞介素1β、CCL-1、CCR-7等16个促炎基因上调,而该作用被亲水改性微粗糙喷砂、酸蚀抵消,与微粗糙喷砂、酸蚀相比,亲水改性微粗糙喷砂、酸蚀表面上肿瘤坏死因子、白细胞介素1α和β等10个基因下调,骨桥蛋白和CCR5上调,在蛋白质水平得到证实。亲水钛表面可调节人类巨噬细胞促炎症细胞因子基因表达和蛋白质分泌,减弱炎症反应,促进骨愈合 。增加粗糙度和润湿性的表面修饰诱导抗炎态(M2样)巨噬细胞活化,表现为白细胞介素4和白细胞介素10水平升高,产生抗炎微环境,引导免疫反应向增进组织愈合方向进行[25]。与疏水性低能量微粗糙喷砂、酸蚀表面相比,亲水性高能量微粗糙喷砂、酸蚀表面的细胞表现为M2激活的抗炎标志物基因表达。钛锆合金比钛的微粗糙喷砂、酸蚀和亲水改性微粗糙喷砂、酸蚀上释放更多的抗炎细胞因子。将高能量和改变的表面化学相组合的钛锆合金亲水改性微粗糙喷砂、酸蚀,能使巨噬细胞产生最大的抗炎微环境并减少促炎因子继续释放[26]。 分别采用大粒颗粒喷砂、酸蚀微糙和亲水改性的酸蚀微糙表面覆盖1型糖尿病大鼠中临界性骨缺损后,可见糖尿病显著增加炎症细胞因子MIP-2和单核细胞趋化蛋白1在骨愈合过程中的表达,亲水改性的酸蚀微糙表面促进钛黏附的M2样巨噬细胞表型反应。在健康和1型糖尿病条件下,亲水性表面均可通过调控早期愈合期内的巨噬细胞表型反应,促进种植体早期骨愈合[27]。亲水表面上观察到血小板附着物的附着降低,微粗糙的钛表面明显减弱血小板释放物对于巨噬细胞的促炎反应;亲水钛表面增强抗炎反应。钛表面形貌和亲水性能够影响血小板释放的蛋白质组学特征,随之影响巨噬细胞促炎细胞因子的表达,这种免疫调节可能是钛表面修饰影响骨整合的重要机制[28]。 羟基磷灰石和磷酸钙对巨噬细胞极化的影响:将人单核/巨噬细胞在具有相似微观粗糙度和不同相组成、纳米级粗糙度和润湿性的无定形或结晶羟基磷灰石涂层上培养,在没有脂多糖刺激条件下,结晶型薄羟基磷灰石涂层引起的炎症反应相对较低[29]。微米结构板状羟基磷灰石晶体基质上培养的巨噬细胞活性氧释放较高,而纳米针状羟基磷灰石基底上几乎不存在活性氧的释放。然而,巨噬细胞增殖的差异更大程度上归因于与基质之间不同的离子交换和蛋白质吸附/保留而非材料质地[30]。纳米CaP修饰表面引起RAW 264.7细胞中诸多促炎症细胞因子和趋化因子基因表达谱的明显下调,Ti-6Al-4V促炎症标志物水平相对较低[31]。 添加金属成分化学改性可激活巨噬细胞M2型转化:Lee等[32]发现,二价阳离子钙和锶(Ca,Sr)化学表面修饰调节J774.A1巨噬细胞黏附形态,明显上调M2巨噬细胞基因型表达。亲水性酸蚀微粗糙种植体表面结合含锶的微米结构钛酸锶,促进兔松质骨中成骨基因Runx2、 osterix和osteocalcin的表达,抑制破骨基因TRAP表达,促进早期骨结合[33]。等离子喷涂的羟基磷灰石涂层结合氧化铈陶瓷颗粒,在促进骨髓间充质干细胞成骨分化的同时诱导巨噬细胞向M2表型极化,表现为M2标志物CD163和CD206的表达上调,抗炎细胞因子(白细胞介素10和白细胞介素1受体拮抗剂)和成骨细胞生成相关基因(骨形态发生蛋白2和转化生长因子β1)升高,M1表面标志物(CCR7和CD11c)和促炎细胞因子(肿瘤坏死因子α和白细胞介素6)下调,活性氧的产生减少,有效促进成骨反应和减少炎症反应[34]。 2.2.2 种植体表面生物改性对巨噬细胞极化的影响 表面修饰嗜中性粒细胞抗菌肽提取物AMP的钛种植体,在脂多糖刺激下巨噬细胞M1极化,NO和超氧化物水平增加;在M2刺激下则精氨酸酶较高,自由基的产生较低,同时表现出促炎和抗炎特征[35]。与未激活的M0巨噬细胞相比,趋化和炎症反应钛颗粒在M1巨噬细胞中显著增强,在M2巨噬细胞中有效抑制,toll样受体信号通路和nod样受体信号通路可能参与该过程[36]。钛表面定植杆菌肽能够提高钛表面亲水性,改善骨髓间充质干细胞的黏附、增殖和成骨分化,同时抑制巨噬细胞扩散或活化和炎性因子的分泌[37]。巨噬细胞的极性可通过整合素介导的人单核细胞THP-1细胞和胶原衍生基质之间的相互作用来控制。缺乏细胞附着序列的生物材料产生促炎M1巨噬细胞,而在白细胞介素4条件下、具有附着位点的材料诱导抗炎表型,促进M2型分化。抑制整合素α2β1阻断M2巨噬细胞的诱导。整合素介导的基于3D微环境的生物材料,编程控制巨噬细胞极性代表了一种新颖、多功能而有效的种植体改性策略[38]。 2.3 种植体表面物理化学性质和生物改性对巨噬细胞诱导成骨的影响 2.3.1 种植体表面物理、化学性质对巨噬细胞诱导成骨的影响 种植体表面纳米形貌对巨噬细胞诱导成骨的影响:Thalji等[39]将适度粗糙表面或超强纳米尺度形貌表面的纯钛种植体分别植入健康受试者的缺牙部位,Affymetrix的人类全基因组1.1ST微阵列功能分类显示在两种表面上,细胞外基质、胶原纤维丝组成、血管化生相关的基因均上调。巨噬细胞替代激活途径相关的基因标志物表达大量上调,包括membrane receptors mannose receptor, C type 1(MRC1)和趋化因子(CCL18、CCL22)等。CCL18在炎症阶段通过上调白细胞介素10抑制Th1细胞因子的产生,在没有白细胞介素4存在的条件下诱导M2谱的巨噬细胞表型。同时发现趋化因子的下调(CCL3、CCL4、CXCL1等),白细胞介素及其相关受体和激酶的下调。种植体表面形貌所导致的基因表达变化涉及细胞外基质组织、免疫调节、血管化生和骨骼重建等诸多方面,激活的M2型巨噬细胞替代活化途径关键标志物和炎性作用的减弱,提示胶原纤维化和免疫调节在改变种植体-骨界面骨量和骨生物力学物理性质、促进骨整合方面的重要作用。 骨髓基质细胞条件培养液中含有增加骨髓单核/巨噬细胞系破骨细胞发生的热敏因子,来自微米和纳米尺度表面黏附的骨髓基质细胞条件培养液增加破骨细胞数。由于破骨细胞在种植体植入后的骨形成阶段介导坏死骨移除,同时参与编织骨的功能性骨重建,加之考虑到表面形貌促进骨形成的因素叠加,破骨细胞数目的一过性增高与总体的骨量增加并不矛盾。因此,种植体黏附细胞在种植体-骨界面发出的局部信号传导,可间接控制骨内种植体周围的破骨细胞形成和骨量,具体机制尚需进一步研究[40]。Ma等[41]发现,巨噬细胞的条件培养液诱导骨髓间充质干细胞的胶原合成和细胞外基质矿化能力,其中30 nm直径较之80 nm直径的纳米管种植体表面显示更多的骨形成、更低的炎症和降低的CD68巨噬细胞分布,80 nm直径纳米管种植体表面诱导明显的多核巨细胞和破骨细胞形成。 种植体表面粗糙度和亲水性对于巨噬细胞诱导成骨的影响:Hefti等[42]发现天然骨表面的破骨吸收小坑(长度、宽度和深度)与先进钛和氧化锆种植体表面特征在尺寸上类似,可作为骨重塑的结构要求与新型种植体表面设计的参考。Brinkmann等[43]研究发现,破骨细胞在粗糙喷砂酸蚀表面上和骨骼上表现出类似的特征,但在光滑钛表面上的活性降低。结果提示在骨重建中,破骨细胞和成骨细胞二者的偶联导致粗糙表面较之光滑表面获得更为成功的骨整合。Milleret等[44]研究以碱处理获得的超亲水微粗糙SBA表面的血小板活化、血块形成结构及对巨噬细胞/单核细胞黏附的影响。研究揭示,增加血液与材料表面接触时间导致更大量的血小板活化、更多的血块形成和更强的补体激活。与钛相比,SBA表面巨噬细胞/单核细胞的数量显著降低,血块聚集呈现超过30 mm厚度的有组织和分层结构(钛表面血块15 mm以下呈斑块状和无结构状,缺乏大的纤维蛋白纤维网络)。 生物材料表面化学性质对巨噬细胞诱导成骨的影响:Mamalis等[45]发现与喷砂、酸蚀表面相比,预粗糙化学改性的活性亲水喷砂、酸蚀表面上生长的人牙周膜细胞hPDL增殖速率和碱性磷酸酶活性增加,骨形态发生蛋白7、Cbfa-1、OPG基因表达显著上调,RANKL基因表达下调,产生一个保护骨骼的微环境,增强骨整合。Davison等[46]发现与微米级β-磷酸三钙的表面相比,亚微米β-磷酸三钙表面结构促进更大、更活跃的破骨细胞分化,诱导人间充质干细胞的成骨细胞分化,通过控制破骨细胞样细胞诱导大量骨形成并达到与之相平衡的吸收速率。具有骨诱导性的双相磷酸钙陶瓷及其降解产物诱导巨噬细胞表达和分泌各种信号分子,招募和促进间充质干细胞分化为成骨细胞。与双相磷酸钙条件培养液相比,降解颗粒发挥了更为实质性的作用。由双相磷酸钙陶瓷及其降解产物引发的炎症反应可能是陶瓷骨诱导所必需的组成部分[47]。简单臭氧气体官能化成功制备的超亲水钛(O3-Ti)促进大鼠骨髓来源间充质干细胞的分化和矿化,极大地减轻促炎症细胞因子产生,不影响RAW264.7巨噬细胞增殖,同时减轻幼稚和脂多糖刺激的巨噬细胞中促进炎症细胞因子产生[48]。 2.3.2 生物材料表面生物改性对巨噬细胞诱导成骨的影响 采用模拟细胞外基质涂层进行表面改性的常用方法包括:胶原蛋白、羟基磷灰石、硫酸软骨素和RGD肽序列[49]。 与Ti针种植体相比,钛/Ⅰ型胶原蛋白针种植体检出数量较多的组织蛋白酶D、骨桥蛋白和骨粘连蛋白阳性细胞。同时,羟基磷灰石与Ⅰ型胶原的组合被认为特别适合骨代谢,可伴有较好的骨细胞附着,由巨噬细胞介导的生物降解和在周围组织中诱导基本的多细胞单位。大鼠胫骨模型显示,羟基磷灰石/Ⅰ型胶原种植体周围可见更多的组织蛋白酶D阳性细胞,只有短暂的炎症反应发生,组织形态计量分析显示:羟基磷灰石/Ⅰ型胶原植入物周围平均骨-种植体骨结合率更高。硫酸软骨素是重要的糖胺聚糖,可见于软骨及松质骨和皮质骨中。大鼠胫骨动物模型显示,在Ⅰ型胶原/硫酸软骨素涂层钛针周围观察到修复性肉芽组织,伴随着植入物周围大量的单核巨噬细胞浸润,显示对组织蛋白酶D免疫反应性巨噬细胞的存在。在Ⅰ型胶原/硫酸软骨素植入物表面新沉积的类骨质和编织骨TRAP阳性破骨细胞数,显著高于Ⅰ型胶原涂层植入物或纯钛种植体。RGD肽序列(Arg-Gly-Asp)是普遍存在的黏附位点,负责细胞整合素受体与细胞外基质的蛋白质互动。RGD和相关肽增强成骨细胞的黏附、迁移和成骨细胞基因表达。RGD涂层后,骨-种植体骨结合率较纯钛种植体明显增强,组织蛋白酶D阳性巨噬细胞提示在钛/RGD针周围发生早期骨重建活动。TRAP阳性破骨细胞大部分出现于植入物表面新沉积的类骨质和编织骨上。结果表明具有RGD序列的种植体表面可直接激活巨噬细胞、成骨细胞和破骨细胞,促进骨形成和骨重建。Nayak等[50]在非桑树来源的花药丝氨酸丝胶蛋白、丝胶蛋白/ RGD固定和原始钛表面上分别进行成骨细胞培养,发现与原始钛相比,表现为成骨细胞的黏附、增殖和分化活力增强,骨涎蛋白、骨钙素和碱性磷酸酶的mRNA表达均上调。此外,当巨噬细胞、成骨细胞-巨噬细胞共培养细胞在丝胶蛋白和丝胶蛋白/ RGD固定的钛表面上培养时,未检测到明显增加的促炎细胞因子肿瘤坏死因子α、白细胞介素1β和一氧化氮的表达。提示丝胶蛋白固定的钛表面是一种具有应用前景的生物活性涂层材料。 将杆菌肽固定于钛表面后获得的亲水性多功能钛种植体,提高人骨髓间充质干细胞的成骨分化,具有优良的抗菌性能。杆菌肽固定化钛可提高骨髓间充质干细胞的黏附、增殖和成骨分化。巨噬细胞难以在杆菌肽固定化钛表面扩散或活化,炎性因子的分泌受抑制[51]。Chen等[52]采用β-磷酸三钙作为模型生物材料来研究巨噬细胞对材料在体外刺激成骨的作用。β-磷酸三钙提取物使得巨噬细胞表型转换到M2极性,其与钙敏感受体(CaSR)途径的激活有关。骨形态发生蛋白2也被β-磷酸三钙刺激显著上调,提示巨噬细胞可能参与β-磷酸三钙刺激的成骨。有趣的是,将巨噬细胞条件的β-磷酸三钙提取物用于骨髓间充质干细胞,骨髓间充质干细胞的成骨分化明显增强,表明巨噬细胞在生物材料诱导成骨中的重要作用。Chen等[11]进一步将β-磷酸三钙涂覆于镁支架上,使得巨噬细胞转换为M2表型,以响应Mg-β-磷酸三钙支架,这可能是由于Toll样受体(TLR)信号通路的抑制作用。 血管内皮生长因子和骨形态发生蛋白2显著上调,提示暴露于Mg-β-磷酸三钙支架的巨噬细胞具有促成骨特性。当骨髓间充质干细胞受到培养在Mg-β-磷酸三钙支架上培养的巨噬细胞条件培养液刺激时,骨髓间充质干细胞向成骨细胞的分化显著增强,而破骨细胞生成受到抑制,表现为MCSF、TRAP下调和RANKL/RANK系统的抑制。结果提示,Mg-β-磷酸三钙支架具有免疫调节特性,可将免疫微环境调控为成骨作用,压倒破骨作用。 "
| [1]Xing H,Wang X,Xiao S,et al.Osseointegration of layer-by-layer polyelectrolyte multilayers loaded with IGF1 and coated on titanium implant under osteoporotic condition.Int J Nanomedicine. 2017;12:7709-7720.[2]Terheyden H,Lang NP,Bierbaum S,et al.Osseointegration - communication of cells.Clin Oral Implants Res.2012;23(10):1127-1135.[3]Sinder BP,Pettit AR,McCauley LK.Macrophages: Their Emerging Roles in Bone.J Bone Miner Res. 2015;30(12):2140-2149.[4]Miron RJ,Bosshardt DD.OsteoMacs: Key players around bone biomaterials. Biomaterials.2016;82:1-19.[5]Davies LC,Jenkins SJ,Allen JE,et al.Tissue-resident macrophages.Nat Immunol.2013;14(10):986-995.[6]Mantovani A,Biswas SK,Galdiero MR,et al.Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol. 2013;229: 176-185.[7]Anton K,Banerjee D,Glod J.Macrophage-associated mesenchymal stem cells assume an activated, migratory, pro-inflammatory phenotype with increased IL-6 and CXCL10 secretion.PloS One. 2012;7: e35036.[8]Mosser DM,Edwards JP.Exploring the full spectrum of macrophage activation.Nat Rev Immunol. 2008;8:958-969. [9]Martinez FO,Gordon S.The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep.2014;6:13. [10]Tugal D,Liao X,Jain MK.Transcriptional control of macrophage polarization. Arterioscler Thromb Vasc Biol.2013;33(6):1135-1144. [11]Chen Z,Mao X,Tan L,et al.Osteoimmunomodulatory properties of magnesium scaffolds coated with β-tricalcium phosphate. Biomaterials. 2014;35(30):8553-8565.[12]Murray PJ,Allen JE,Biswas SK,et al.Macrophage activation and polarization: nomenclature and experimental guidelines.Immunity. 2014;41:14-20. [13]Ziegler T,Rausch S,Steinfelder S,et al.A novel regulatory macrophage induced by a helminth molecule instructs IL-10 in CD4+ T cells and protects against mucosal inflammation.J Immunol. 2015;194(4): 1555-1564.[14]Trindade R,Albrektsson T,Wennerberg A.Current concepts for the biological basis of dental implants: foreign body equilibrium and osseointegration dynamics.Oral Maxillofac Surg Clin North Am. 2015;27(2):175-183. [15]Gibon E,Lu LY,Nathan K,et al.Inflammation, ageing, and bone regeneration.J Orthop Translat. 2017;10:28-35. [16]Cho SW,Soki FN,Koh AJ,et al.Osteal macrophages support physiologic skeletal remodeling and anabolic actions of parathyroid hormone in bone.Proc Natl Acad Sci U S A.2014;111(4):1545-1550.[17]Guihard P,Danger Y,Brounais B,et al.Induction of osteogenesis in mesenchymal stem cells by activated monocytes/macrophages depends on oncostatin M signaling.Stem Cells.2012;30(4):762-772.[18]Karazisis D,Ballo AM,Petronis S,et al.The role of well-defined nanotopography of titanium implants on osseointegration: cellular and molecular events in vivo.Int J Nanomedicine.2016;11:1367-1382. [19]Karazisis D,Petronis S,Agheli H,et al.The influence of controlled surface nanotopography on the early biological events of osseointegration. Acta Biomater.2017;53:559-571.[20]Lü WL,Wang N,Gao P,et al.Effects of anodic titanium dioxide nanotubes of different diameters on macrophage secretion and expression of cytokines and chemokines.Cell Prolif.2015;48(1):95-104.[21]Ma QL, Zhao LZ, Liu RR, et al. Improved implant osseointegration of a nanostructured titanium surface via mediation of macrophage polarization.Biomaterials.2014;35(37):9853-9867. [22]樊牮,邹耿森,陈江.钛种植体表面纳米改性及其与机体免疫应答[J].国际口腔医学杂志, 2014,41(6):691-693.[23]He XT,Li X,Yin Y,et al.The effects of conditioned media generated by polarized macrophages on the cellular behaviours of bone marrow mesenchymal stem cells.J Cell Mol Med.2017.doi: 10.1111/jcmm.13431.[Epub ahead of print][24]Alfarsi MA,Hamlet SM,Ivanovski S.Titanium surface hydrophilicity modulates the human macrophage inflammatorycytokine response.J Biomed Mater Res A.2014;102(1):60-67. [25]Hotchkiss KM,Reddy GB,Hyzy SL,et al.Titanium surface characteristics, including topography and wettability, alter macrophage activation.Acta Biomater.2016;31:425-434. [26]Hotchkiss KM,Ayad NB,Hyzy SL,et al.Dental implant surface chemistry and energy alter macrophage activation in vitro.Clin Oral Implants Res.2017;28(4):414-423.[27]Lee RS,Hamlet SM,Ivanovski S,et al.The influence of titanium surface characteristics on macrophage phenotype polarization during osseous healing in type I diabetic rats: a pilot study.Clin Oral Implants Res. 2017;28(10):e159-e168. [28]Alfarsi MA,Hamlet SM,Ivanovski S.The Effect of Platelet Proteins Released in Response to Titanium Implant Surfaces on Macrophage Pro-Inflammatory Cytokine Gene Expression. Clin Implant Dent Relat Res. 2015;17(6):1036-1047. [29]Rydén L,Omar O,Johansson A,et al.Inflammatory cell response to ultra-thin amorphous and crystalline hydroxyapatite surfaces.J Mater Sci Mater Med.2017;28(1):9.[30]Mestres G,Espanol M,Xia W,et al.Inflammatory response to nano- and microstructured hydroxyapatite. PLoS One.2015;10(3):e0120381. [31]Hamlet S,Ivanovski S.Inflammatory cytokine response to titanium chemical composition and nanoscale calcium phosphate surface modification.Acta Biomater.2011;7(5):234523-234553. [32]Lee CH,Kim YJ,Jang JH,et al.Modulating macrophage polarization with divalent cations in nanostructured titanium implant surfaces. Nanotechnology.2016;27(8):085101. [33]Park JW,Kim YJ,Jang JH,et al.Positive modulation of osteogenesis- and osteoclastogenesis-related gene expression with strontium-containing microstructured Ti implants in rabbit cancellous bone.J Biomed Mater Res A.2013;101(1):298-306.[34]Li K,Shen Q,Xie Y,et al.Incorporation of cerium oxide into hydroxyapatite coating regulates osteogenic activity of mesenchymal stem cell and macrophage polarization.J Biomater Appl. 2017;31(7): 1062-1076.[35]Wessely-Szponder J,Szponder T,Bobowiec R.Different activation of monocyte-derived macrophages by antimicrobial peptides at a titanium tibial implantation in rabbits.Res Vet Sci.2017;115:201-210.[36]Pajarinen J,Kouri VP,Jämsen E,et al.The response of macrophages to titanium particles is determined by macrophage polarization.Acta Biomater.2013;9(11):9229-9240. [37]Nie B,Ao H,Zhou J,et al.Biofunctionalization of titanium with bacitracin immobilization shows potential for anti-bacteria, osteogenesis and reduction of macrophage inflammation.Colloids Surf B Biointerfaces. 2016;145:728-739. [38]Cha BH,Shin SR,Leijten J,et al.Integrin-Mediated Interactions Control Macrophage Polarization in 3D Hydrogels.Adv Healthc Mater. 2017;6(21).doi: 10.1002/adhm.201700289.Epub 2017 Aug 7.[39]Thalji GN,Nares S,Cooper LF.Early molecular assessment of osseointegration in humans. Clin Oral Implants Res.2014;25(11):1273-1285. [40]Nagasawa M,Cooper LF,Ogino Y,et al.Topography Influences Adherent Cell Regulation of Osteoclastogenesis.J Dent Res. 2016; 95(3):319-326. [41]Ma QL,Fang L,Jiang N,et al.Bone mesenchymal stem cell secretion of sRANKL/OPG/M-CSF in response to macrophage-mediated inflammatory response influences osteogenesis on nanostructured Ti surfaces. Biomaterials.2017;154:234-247.[42]Hefti T,Frischherz M,Spencer ND,et al.A comparison of osteoclast resorption pits on bone with titanium and zirconia surfaces. Biomaterials. 2010;31(28):7321-7331.[43]Brinkmann J,Hefti T,Schlottig F,et al.Response of osteoclasts to titanium surfaces with increasing surface roughness: an in vitro study.Biointerphases.2012;7(1-4):34. [44]Milleret V,Tugulu S,Schlottig F,et al.Alkali treatment of microrough titanium surfaces affects macrophage/monocyte adhesion, platelet activation and architecture of blood clot formation. Eur Cell Mater. 2011;21:430-444;discussion 444.[45]Mamalis AA,Markopoulou C,Vrotsos I,et al.Chemical modification of an implant surface increases osteogenesis and simultaneously reduces osteoclastogenesis: an in vitro study.Clin Oral Implants Res. 2011;22(6):619-626. [46]Davison NL,Luo X,Schoenmaker T,et al.Submicron-scale surface architecture of tricalcium phosphate directs osteogenesis in vitro and in vivo.Eur Cell Mater.2014;27:281-297;discussion 296-297.[47]Wang J,Liu D,Guo B,et al.Role of biphasic calcium phosphate ceramic-mediated secretion of signaling molecules by macrophages in migration and osteoblastic differentiation of MSCs.Acta Biomater. 2017;51:447-460. [48]Sunarso,Toita R,Tsuru K,et al.A superhydrophilic titanium implant functionalized by ozone gas modulates bone marrow cell and macrophage responses.J Mater Sci Mater Med.2016;27(8):127.[49]Förster Y,Rentsch C,Schneiders W,et al.Surface modification of implants in long bone.Biomatter. 2012;2(3):149-157.[50]Nayak S,Dey T,Naskar D,et al.The promotion of osseointegration of titanium surfaces by coating with silk protein sericin.Biomaterials. 2013;34(12):2855-2864.[51]Nie B,Ao H,Zhou J,et al.Biofunctionalization of titanium with bacitracin immobilization shows potential for anti-bacteria, osteogenesis and reduction of macrophage inflammation.Colloids Surf B Biointerfaces. 2016;145:728-739. [52]Chen Z,Wu C,Gu W,et al.Osteogenic differentiation of bone marrow MSCs by β-tricalcium phosphate stimulating macrophages via BMP2 signalling pathway.Biomaterials.2014;35(5):1507-1518.[53]Miron R,Bosshardt D.Multi-Nucleated Giant Cells: Good Guys or Bad Guys? Tissue Eng Part B Rev. 2017.doi:10.1089/ten.TEB.2017.0242.[Epub ahead of print] [54]Thalji G,Cooper LF.Molecular assessment of osseointegration in vitro: a review of current literature.Int J Oral Maxillofac Implants. 2014; 29(2):e171-199. [55]Wythe SE,Nicolaidou V,Horwood NJ.Cells of the immune system orchestrate changes in bone cell function.Calcif Tissue Int. 2014; 94(1):98-111. [56]Takayanagi H.New developments in osteoimmunology.Nat Rev Rheumatol.2012;8(11):684-689.[57]Trindade R,Albrektsson T,Tengvall P,et al.Foreign Body Reaction to Biomaterials: On Mechanisms for Buildup and Breakdown of Osseointegration.Clin Implant Dent Relat Res.2016;18(1):192-203. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [5] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [6] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Cheng Jun, Tan Jun, Zhao Yun, Cheng Fangdong, Shi Guojia. Effect of thrombin concentration on the prevention of postoperative cerebrospinal leakage by fibrin glue [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 570-575. |
| [9] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [10] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [11] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [12] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [13] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [14] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [15] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||